By Huanfen Yao, Ilkka Lähdesmäki & Babak A. Parviz
For the past number of years diabetes mellitus has gained importance globally as a major public heath concern. The chronic high glucose levels in blood can be a further risk for other serious micro-vascular complications, such as stroke, atherosclerosis, and coronary artery diseases. The data provided by the International Diabetes Federation shows that, for the year 2010, more than 6% of the world population suffered from diabetes mellitus.
Self-monitoring of blood glucose is widely used for controlling glucose levels. This is usually performed by a “finger prick” test using a portable meter. However, this sensing method is invasive; it requires direct contact between the sensor and the blood. More importantly, it can provide only a single temporal value – it cannot follow the rapid fluctuations of blood glucose levels during diabetes medication. Hence, a noninvasive continuous glucose sensing method would be strongly preferred for the next generation of human health monitoring.
Non-invasive detection is usually achieved by placing the sensing element on the skin, and sensing tissue glucose without piercing the skin, using techniques such as absorption spectrometry and polarimetry. But these methods require both the sensor and various auxiliary components for practical operation, which make them less portable. An alternative way of non-invasive sensing can be achieved by taking measurements from body fluids, such as urine, saliva, sweat or tear fluid. Besides the drawback of the variable water content, or dilution effect, urine cannot be accessed in a continuous basis, and placing sensors in the mouth is uncomfortable. Compared with the difficulty encountered in sweat harvest, tear fluid is directly accessible on the eye and can provide a unique interface between the sensor and the body. As reported in prior clinical studies, there is a correlation between the tear glucose and the blood glucose, for both nondiabetic and diabetic groups.
Our group aims to build a contact lens complete with sensors and radio to sample the tear fluid, perform an analysis, and wirelessly transmit the results. The contact lens allows for monitoring human health non-invasively and continuously through the tear fluid. This contact lens can be built on a polymer platform, complete with a biosensor module, communication circuit, antenna and embedded interconnects, leaving the central area clear for the visual path (Fig. 1). Sensor placement in tear fluid permits the use of continuous sensing mode, which would provide a more complete understanding of the human health status. The contact lens may have the potential benefit of eliminating the finger prick test and increase compliance with glucose monitoring among diabetic patients.
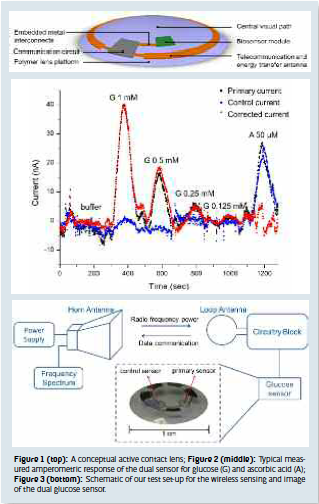
Challenges for glucose sensor design include the need for enhanced sensitivity and interference rejection, especially for glucose measurement in the tear fluid, because the glucose levels in tears are much lower (0.1~0.6 mM) compared with blood concentration (4~6 mM), and there are various interfering molecules, such as ascorbic acid, lactate, and urea present on the surface of the eye. Also, different proteins could influence the sensor output. As a result, detection requirements for a tear glucose sensor are more stringent than those for a blood glucose sensor. In our project, we chose an electrochemical sensing method to analyze the tear fluid due to its real-time, fast, and easy operation. We developed a dual microscale sensor design for interference compensation (Fig. 3), where the primary sensor responds to glucose while both the primary and the control sensor respond equally to the interferences. Different response behavior on the primary and the control sensor is due to different surface treatment. When a difference signal is calculated, the interference response is removed while the glucose response is preserved. We have fabricated such a dual sensor on a PET (polyethylene terephthalate) substrate and demonstrated that it can be successfully used for sensing low concentrations of glucose in the presence of interfering chemical ascorbic acid (Fig. 2).
Another challenge for developing the active contact lens is the construction of the wireless power transmission and readout circuitry parts to support the sensor. Figure 3 shows the wireless sensing system setup which we developed for test and characterization. For measurement, we used a horn antenna to broadcast radio waves towards a custom-designed radio chip designed at the University of Washington. A loop antenna, which can be fabricated on the contact lens, is used to collect the radio frequency wave power and supply it to the circuitry. The same loop antenna can also transmit data, in form of a shift in peak frequency. The current signal from the sensor is converted to a shift in the frequency spectrum through the readout circuit. Thus, glucose concentration data can be transferred from the sensor to an external storage unit as frequency spectra wirelessly.
At this stage, we have managed to fabricate the circuitry part in a small chip of 0.5 mm2, which provides a potential to construct the glucose sensor and supporting circuit on a single contact lens in the future. We have also obtained linear measurement results for glucose up to 2 mM, by reading the frequency shift using the wireless sensing method. Combining the dual sensor and the communication circuitry part, we hope to build a wireless non-invasive continuous glucose sensor in the future.
Our active contact lens is still under development and far from complete. Future improvements will include more biocompatible encapsulation, building low noise interfaces, and complete component integration. We believe that an integrated contact lens could be the next generation system to monitor several physiological and metabolic indicators for human health, in a wireless, non-invasive and continuous way.
Huanfen Yao, MSc Ilkka Lähdesmäki, Ph.D. Babak A. Parviz, Ph.D. University of Washington U.S.A. parviz@u.washington.edu
About Brenda Wiederhold
President of Virtual Reality Medical Institute (VRMI) in Brussels, Belgium.
Executive VP Virtual Reality Medical Center (VRMC), based in San Diego and Los Angeles, California.
CEO of Interactive Media Institute a 501c3 non-profit
Clinical Instructor in Department of Psychiatry at UCSD
Founder of CyberPsychology, CyberTherapy, & Social Networking Conference
Visiting Professor at Catholic University Milan.








